Photogenerated charge transfer is an important phenomenon in photochemistry and photophysics. Especially in the catalytic chemical reactions involving photogenerated charges, the utilization rate of photogenerated carriers greatly affects or even determines the catalytic reaction rate. Facing the shortage of high-efficiency light-harvesting materials and the generally low photon yield, how to optimize and improve the separation and transfer speed of photogenerated charges urgently needs to be cracked by new methods, new theories, and new technologies.
In recent years, the new way of modulating photogenerated charge transfer by the piezoelectric polarization effect has shown broad development and application prospects in photocatalytic hydrogen production, carbon dioxide reduction, and organics degradation. At the same time, this strategy has also transformed environmental mechanical energy into chemical energy beecome practical and easy. It should be pointed out that the current theoretical methods and practices for regulating photogenerated charge transfer based on piezoelectric effect still need to be further improved. It is against this background that the research group of Professor Zhai Jiwei in Tongji University, used the solvothermal method to synthesize BiOX/BTO nanocomposites with good photoelectric properties and piezoelectric response properties. The study found that there are two main reasons for this composite material to improve the photocatalytic reaction rate: first, the composite material has formed a large built-in electric field due to the establishment of a new Fermi level balance, and second, the composite material has shown a significantly enhanced piezoelectric potential difference. Mediated by the difference between electrochemical potential and piezoelectric potential, the material exhibits ultra-fast photocatalytic degradation of organics. On the contrary, the catalytic performance of the composite material is greatly reduced under the action of separate ultrasonic or light mediated. This further illustrates the importance of the synergistic effect of piezoelectric potential and photocurrent density in promoting the rate of photocatalytic reaction. The facts show that with BiOBr/BTO as the catalyst, the piezoelectric photocatalytic degradation rate of methyl orange dye molecules is 13.65 and 13.09 times that of photocatalytic and piezoelectric catalytic degradation, respectively. In addition, under the action of external stress, the theoretical piezoelectric potential difference exhibited by BiOBr/BTO composite is as high as 100 mV, which is much larger than that of pure BiOBr nanoplate of 30.0 mV and BTO nanoparticle of 31.2 mV.
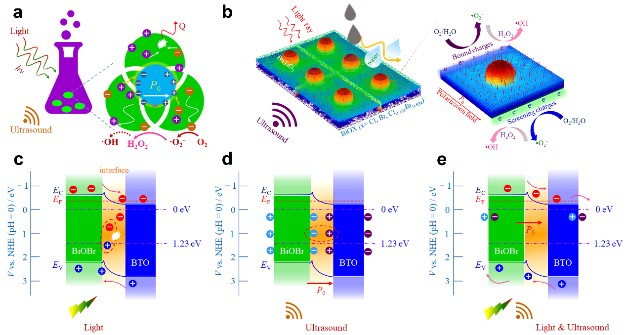
Figure 1. a,b) Schematic illustrating of the simultaneous light and ultrasound-activated piezoelectric composites. c-e) Diagram of the charge transfer mechanism of BiOBr/BTO composites under light illumination, ultrasonic excitation, and light concurrent ultrasonic co-irradiation.
This study provides a new method for further understanding of the internal relationship between piezoelectric potential, electrochemical potential, and photocatalytic chemical reaction rate. The related results were recently published online in Small under the title of "Remarkable Piezophoto Coupling Catalysis Behavior of BiOX/BaTiO3 (X = Cl, Br, Cl0.166Br0.834) Piezoelectric Composites".
Paper link: https://onlinelibrary.wiley.com/doi/10.1002/smll.202001573